|
Noble gases have eight electrons in their outermost shell, except in the case of helium, which has two. Neon, like all noble gases, has a full valence shell. Group 13 has three valence electrons, Group 14 has four, up through Group 18 with eight. What is the valence shell electron configuration of all the noble gases except for helium? In the p block, the number of valence electrons is equal to the group number minus ten. Its electron configuration is (1s^2 2s^2 2p^6 3s^1). In the periodic table, the elements are listed in order of increasing atomic number Z. Sodium, element number eleven, is the first element in the third period of the periodic table. The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. Likewise, people ask, what is the electron configuration for noble gases? Helium, like the other noble gases, is chemically inert. T/F: An oxygen atom, having electron configuration 1s 2s 2p, forms a single bond with another oxygen atom. Helium has monatomic molecules, and is the lightest of all gases except hydrogen. For facts, physical properties, chemical properties, structure and atomic properties of the specific element, click on the element symbol in the below periodic table. In the below periodic table you can see the trend of Valence Electrons. Likewise, do all noble gases have the same valence electron configuration? T/F: All noble gases have the same valence electron configuration. Periodic Table of Elements with Valence Electrons Trends. This is the most stable arrangement of electrons, so noble gases rarely react with other elements and form compounds. That's because they have eight valence electrons, which fill their outer energy level. 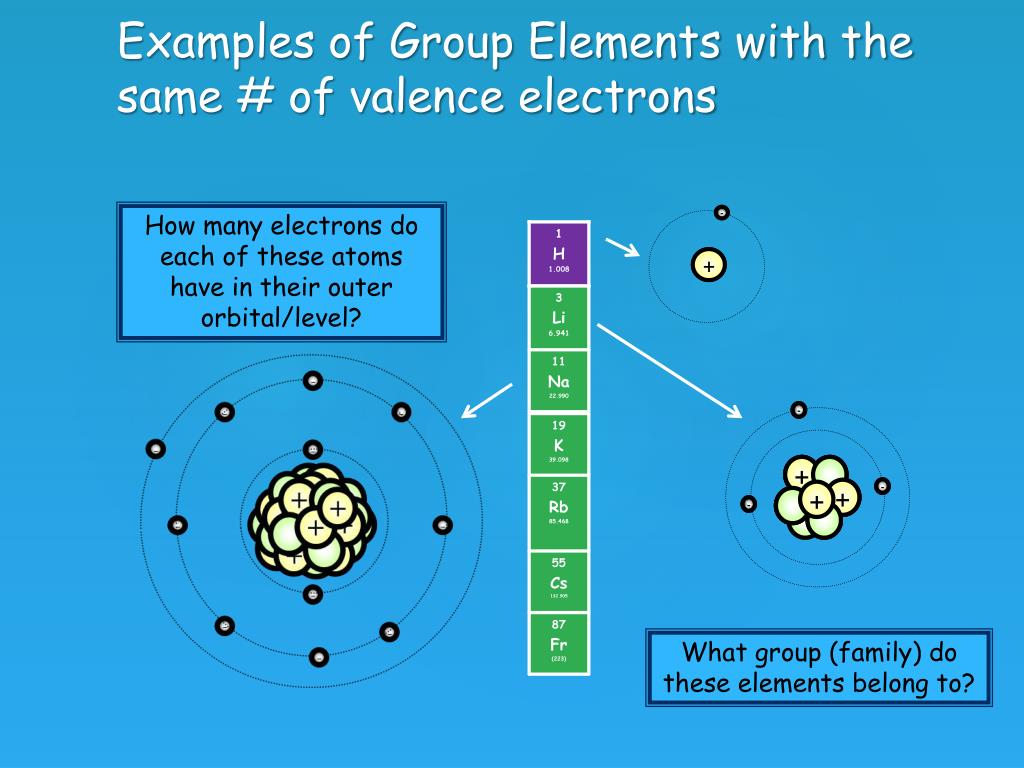
Noble gases are the least reactive of all elements. Likewise, people ask, what are the valence electrons of noble gases? Ok but how many valence electrons does an atom of. The noble gases are the only non-transition-metal elements that have eight valence electrons in their neutral, ground state configuration, but atoms of other elements can produce a full octet - an outer shell with eight electrons - by gaining or losing electrons. How to create helium electron systems using a combination of lithium, potassium, cobalt and iron electrodes, and make a battery.The goal of this article is to help you create your own helium electron system.1.The basic ideaFirst, you need to figure out which electrolytes you are using for your battery. A valence electron is an outer shell electron and may participate in the formation of a chemical bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
